Third Party Manufacturing
Third Party Manufacturing Specification
- Molecular Formula
- As per formulation
- Application
- Hospital, Clinic
- Purity
- 99%
- Grade
- A
- Formulations Type
- General Drugs
- Formulations Form
- Tablets
- Treatments & Functions
- Used for various therapeutic purposes
- Gender/Age Group
- Adult
- Dosage Guidelines
- As prescribed by physician
- Volume
- 500-1000 Tablets Per Pack
- Storage Instructions
- Store in a cool, dry place
- Manufacturer Type
- Third Party Manufacturer
- Packaging Type
- Blister/Alu-Alu/Strip Packaging
- Quality Standard
- WHO-GMP Certified
- Prescription/Non prescription
- Prescription
- Shelf Life
- 24 Months
Third Party Manufacturing Trade Information
- Main Domestic Market
- All India
About Third Party Manufacturing
Third party or contract manufacturing is referred to the outsourcing of pharmaceutical products or to get products manufactured from others manufacturing units with your own brand names. Even, pharma companies having their own manufacturing plants are gotten manufactured their pharma products from other manufacturers.Expert Third-Party Manufacturing
We specialize in the third-party manufacturing of general drug formulations, delivering exceptional product quality and consistency for our clients. Our manufacturing facilities adhere strictly to WHO-GMP certification, ensuring all products meet international healthcare standards. Blister, alu-alu, and strip packaging help optimize product longevity and maintain tablet integrity, making our services highly reliable for hospitals and clinics alike.
Versatile Tablet Formulations
Designed for adult use, our tablet forms are suitable for a wide range of therapeutic purposes. By offering customizable quantity packs (5001000 tablets), we make bulk supply straightforward for clinical and hospital settings. The formulations are handled with meticulous care, ensuring precise dosage, exceptional purity, and robust stability over their extended 24-month shelf life.
FAQs of Third Party Manufacturing:
Q: How does the third-party manufacturing process work for general drug tablets?
A: The third-party manufacturing process involves producing pharmaceuticals on behalf of other companies. We manage the production, quality control, and packaging of general drug tablets, ensuring compliance with WHO-GMP standards. Clients simply provide specifications, and we handle the rest, delivering the finished product in secure, high-quality packaging.Q: What are the benefits of choosing blister, alu-alu, or strip packaging?
A: These packaging options offer excellent protection against moisture, contamination, and physical damage, which helps extend the 24-month shelf life of the tablets. They also make dispensing and dosage tracking more convenient for healthcare professionals and patients alike.Q: When should the tablets be administered, and how is dosage determined?
A: Tablets should be administered strictly according to the dosage guidelines provided by a qualified physician. Each patients condition is unique, so the dosage and timing will be tailored to their specific therapeutic needs, as outlined in the prescription.Q: Where are these tablets intended to be used?
A: Our manufactured tablets are primarily intended for use in hospitals and clinics. Their formulation and volume per pack are optimized for professional healthcare environments where high standards of quality and consistency are required.Q: What quality standards and certifications are maintained during production?
A: All manufacturing processes strictly adhere to WHO-GMP certification requirements. This ensures that products consistently meet high standards of purity, safety, and efficacy throughout their lifecycle.Q: How should these tablets be stored to maintain their quality and shelf life?
A: To ensure optimal stability and effectiveness, tablets should be stored in a cool, dry place away from direct sunlight. Proper storage conditions help preserve the 24-month shelf life and maintain the tablets 99% purity.
- Main Domestic Market
- All India
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Third Party Manufacturing Category
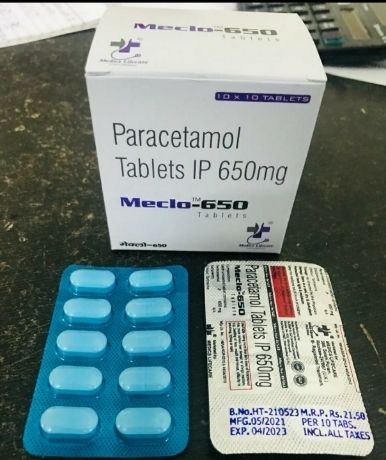
MECLO 650
Volume : 10x10 Tablets per box
Purity : 99%
Dosage Guidelines : As prescribed by physician
Storage Instructions : other, Keep in cool and dry place
FEXOFENADINE AND MONTELUKAST TABLETS
Volume : 10 tablets per blister pack
Purity : Pharmaceutical Grade
Dosage Guidelines : As prescribed by the physician typically one tablet daily
Storage Instructions : other, Store in a cool dry place below 25C. Protect from moisture and direct sunlight.